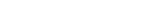IRT
Durchgeführte Studien
in 80 Ländern
Mittlere Mängelzahl bei Akzeptanztests
bei anvisierten ≤ 5 mäßigen
und kleineren Mängeln
Termingerechte Bereitstellung
zur Kundenabnahme
Komplexe klinische Studien? Lernen Sie den umfangreichen Funktionsumfang unseres IRT-Systems kennen. Damit können Sie individuelle Anforderungen erfüllen – ebenso wie enge Zeitpläne.
Die wesentlichen IRT-Kernfunktionen bilden die solide Grundlage. Jede einzelne Funktion ist darauf ausgerichtet, die Anforderungen gängiger Protokolle in komplexen dezentralen, patientenzentrierten Studien für Therapiebereiche wie Onkologie, Zentralnervensystem und seltene Krankheiten zu erfüllen. Des Weiteren können Sie mit den Tools bei Bedarf neue, kundenspezifische Funktionen erstellen. Hierzu stehen leicht zu kombinierende Bausteine bereit. Damit können unsere Serviceteams Sie bei den individuellen Anforderungen jeder Studie unterstützen.
Unser IRT-System bietet jedoch nicht nur eine fortschrittliche Architektur, die einen schnellen und umfassenden Einsatz ermöglicht, sondern auch umfangreiche Konfigurationsmöglichkeiten. Auf diese Weise können Studienteams die Systemfunktionen an die sich ständig ändernden Anforderungen ihrer Studie anpassen. Denn keine Studie unserer Kunden gleicht der anderen. Was sie aber alle gemein haben, sind die Komplexität und der Zeitdruck.
Studienlogistik
-
Rollen, Berechtigungen und Verblindungsmanagement
-
Studien- und Prüfzentrenverwaltung
-
Kohorten-, Abschnitts- und Phasenmanagement
-
Dynamisches Kohorten- und Dosismanagement
-
Zeitplanfunktion für zusätzliche und dynamische Besuchstermine
Patientenlogistik
-
Teilnehmermanagement
-
Adaptive Funktionen für Nachrücker und Randomisierung
-
Crossover-Designs und Wiederholungsbehandlungen
-
Offene Verlängerung
-
Dosisberechnung
-
Teilnehmer-Rollover
-
Dosisanpassung und Behandlungsunterbrechung
Arzneimittellogistik
-
Management der Arzneimittelausgabe
-
Management der Arzneimittelversorgung
-
Arzneimitteldokumentation, Arzneimittelabgleich und Management der Arzneimittelrückgabe und -vernichtung
-
Management von Temperaturabweichungen
-
Management von kontrollierten Substanzen
-
Variable Arzneimittelbeschaffung
-
Zentralapotheke
-
Direktversand an Patienten
-
Robustes Management der Versorgungsstrategie
Das IRT-System wurde für das Unbekannte entwickelt, denn es sollte genauso bereit für Veränderungen sein wie Sie selbst.
Der Forschungsprozess ist nicht statisch, Und ebenso wenig ist es Ihre Studie. Für möglichst umfangreiche Daten müssen Prüfpläne geändert werden, sobald neue Informationen vorliegen. Deshalb haben wir von Suvoda das IRT-System so konzipiert, dass es sich bei Änderungen während einer bereits laufenden Studie bequem anpassen lässt.
Nach der Implementierung können häufig erforderliche Ergänzungen, Modifikationen und Korrekturen in Bezug auf die Studie, die Prüfzentren, das Arzneimittelmanagement und administrative Funktionen von berechtigten Anwendern innerhalb unseres Systems vorgenommen werden. Damit liegt die Kontrolle in Ihren Händen, und der Zeitaufwand und die Kosten für Änderungsaufträge werden reduziert. Auch wenn umfangreichere Aktualisierungen erforderlich sind, kann Ihr dediziertes Suvoda-Projektteam dank unserer modularen Architektur einfach und schnell neue IRT-Funktionen hinzufügen.
Das anwenderfreundliche IRT-System erleichtert es Anwendern aller Erfahrungsstufen, zielgerichtete Maßnahmen zu ergreifen.
Wir haben festgestellt, dass Menschen eher ihr Bestes geben, wenn sie ihre bevorzugten Tools verwenden können.
Deshalb ist das Interagieren mit dem IRT-System von Suvoda unabhängig von der Erfahrung mit Technologien für klinische Studien durchweg einfach gehalten, damit nicht nur das dedizierte Fachpersonal, sondern auch Gelegenheitsanwender am Prüfzentrum und ambulantes Pflegepersonal sich leicht zurechtfinden. Möglich macht dies die Kombination aus schnörkelloser Optik, intuitiver Navigation und patientenzentriertem Arbeitsablauf. So bleibt der Fokus stets auf der unmittelbar vorliegenden Aufgabe.
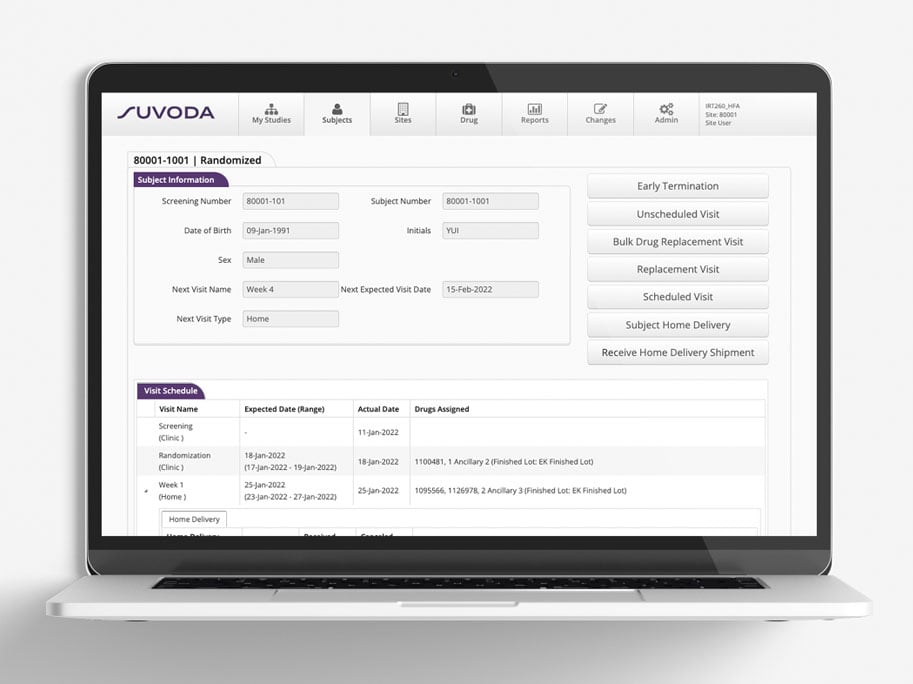
Mit voreingestellten Berichten, Ad-hoc-Berichten sowie erweiterten Analysen stets up to date.
Die voreingestellten Berichte und Ad-hoc-Berichte des IRT-Systems von Suvoda liefern Ihnen minutengenaue Darstellungen von Datenpunkten, Leistungskennzahlen (Key Performance Indicators, KPIs) und Trends zu Teilnehmern, Prüfzentren, Arzneimitteln und Depots innerhalb der jeweiligen Studie. So wissen Sie immer, wo Sie stehen und was Ihrer Aufmerksamkeit bedarf.
Warum eine derart robuste Berichterstellung? Damit erhalten Sie die erforderlichen Informationen, die Sie für eine bessere Entscheidungsfindung bei der Durchführung der Studien von heute – und der Planung der Studien von morgen – benötigen.
AUSGEWÄHLTE IRT-MODULE

Volle Kontrolle über die Kühlkette